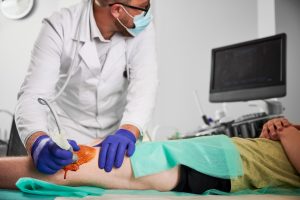
Chronic joint pain has a way of narrowing your world. Steps you once took without thinking become calculated decisions, and the fear of surgery looms over every conversation with your doctor. But surgery is not your only option. Stem cell therapy has emerged as a scientifically grounded, non-surgical approach to managing joint pain and degeneration, offering real hope for adults who want to stay active without going under the knife. This guide walks you through exactly how it works, what the research says, who benefits most, and what to realistically expect from treatment.
Table of Contents
- What is stem cell therapy for joints?
- How does stem cell therapy work for joint pain?
- What does the research say about effectiveness and safety?
- Nuances: Who benefits, edge cases, and what to ask your provider
- The real story: What most people misunderstand about stem cell therapy for joints
- Explore your options for joint pain recovery
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Not a cure-all | Stem cell therapy often eases joint pain and improves movement, but rarely rebuilds cartilage. |
| Most see symptom improvement | Many patients experience modest pain and function benefits, lasting up to a year. |
| Generally safe option | Side effects are usually mild and serious risks are rare in studies. |
| Best after other therapies | Stem cell therapy suits those not helped by physical therapy or standard injections. |
What is stem cell therapy for joints?
Let’s start with a clear definition. Stem cell therapy for joints involves injecting specialized cells, most commonly mesenchymal stem cells (MSCs), directly into a damaged or degenerated joint. These cells have a unique ability to communicate with surrounding tissue, reduce inflammation, and support the environment needed for repair.
MSCs come from three primary sources, each with its own practical advantages:
- Bone marrow (BM-MSCs): Harvested from the patient’s own hip or pelvis, these cells have the longest track record in orthopedic research and tend to show consistent results in clinical trials.
- Adipose tissue (AD-MSCs): Collected from fat tissue, usually through a minor liposuction procedure, these cells are abundant and easier to harvest in large quantities.
- Umbilical cord: Sourced from donated cord tissue after birth, these are allogeneic (from another person) and available off the shelf, making them a convenient option for patients who prefer not to undergo a harvesting procedure.
Once injected into the joint, how stem cell therapy works involves a process called paracrine signaling. Rather than simply becoming new cartilage cells outright, MSCs release growth factors and anti-inflammatory proteins that calm the joint environment, support existing tissue, and may encourage the body’s own repair mechanisms to activate. They can also differentiate into chondrocytes, which are the cells responsible for cartilage production, though this is not the primary driver of symptom relief in most cases.
According to Frontiers in Bioengineering & Biotechnology, MSCs from bone marrow, adipose tissue, or umbilical cord are pursued as a non-surgical regenerative option for chronic joint pain because they can differentiate into chondrocytes, release anti-inflammatory factors, modulate immune responses, and promote cartilage repair via paracrine signaling.
This is an outpatient procedure. You arrive, receive a local anesthetic, and the injection is administered with imaging guidance in most cases. There’s no general anesthesia, no lengthy hospital stay, and no surgical incision.
Pro Tip: Ask your provider which cell source they use and why. The choice between autologous (your own cells) and allogeneic (donor cells) affects both the procedure and the potential outcomes for your specific condition.
How does stem cell therapy work for joint pain?
With the basics in mind, let’s look at how stem cell therapy translates into results for joint pain and mobility.
The pain relief mechanism is twofold. First, MSCs release cytokines and growth factors that suppress inflammatory signaling in the joint. Chronic joint pain, particularly from osteoarthritis (OA), is heavily driven by ongoing inflammation that breaks down cartilage over time. By calming that inflammatory cycle, MSCs can reduce the pain signals reaching your brain. Second, the cells may stimulate local repair cells already present in the joint, essentially waking up a healing process that has stalled.
“Meta-analyses show modest but meaningful pain relief and function improvement compared to controls at 6 to 12 months. Patients report better scores on standardized pain and activity measures, though structural changes on MRI are not consistently observed.” — Cochrane Systematic Review
Clinical data supports a measured but real benefit. A Cochrane Systematic Review found that pain scores improved meaningfully, with KOOS pain scores (a standardized knee pain measurement) improving by an average of 15.37 points and daily activity scores improving by 12.84 points compared to control groups at 6 to 12 months.

Here’s a summary of key outcome data:
| Outcome measure | Average improvement | Time frame |
|---|---|---|
| VAS pain score (visual analog scale) | 1.67 to 3.55 point reduction | 6 to 12 months |
| KOOS pain subscale | +15.37 points vs. control | 6 to 12 months |
| KOOS daily activity (ADL) | +12.84 points vs. control | 6 to 12 months |
| MRI structural change | No consistent change observed | Up to 12 months |
These numbers matter because they reflect real-world function. A 15-point improvement on the KOOS scale means walking to the mailbox hurts less, climbing stairs feels safer, and sleep improves because nighttime aching decreases.
It’s also worth noting that placebo effects play a role in any injection-based treatment, contributing to roughly 50 to 63% of perceived improvement in some studies. This is a normal part of how pain management works and does not diminish the genuine benefit. Comparing PRP versus stem cell therapy can help you understand where each option fits in a broader treatment plan.
The honest takeaway: stem cell therapy is not a miraculous cure. It is a clinically supported option for meaningful pain and function improvement, particularly for people who have not found adequate relief from conventional conservative treatments.
What does the research say about effectiveness and safety?
Understanding the mechanism is one thing, but what does the research actually show about effectiveness and safety compared to other treatments?
The procedure itself is straightforward. According to Stem Cell Research & Therapy, the standard approach involves intra-articular injection of MSCs or stromal vascular fraction (SVF) directly into the joint, often as a single dose of 25 million cells or fewer, performed as an outpatient procedure under local anesthetic. Autologous sources (bone marrow or fat) and allogeneic sources are both used depending on the clinical setting and patient needs.
Here’s what a typical treatment experience looks like, step by step:
- Initial consultation and imaging: Your provider reviews your X-rays or MRI to confirm the degree of joint degeneration and determine whether you’re a good candidate.
- Cell sourcing (if autologous): A small amount of bone marrow or fat tissue is collected from your own body using a minimally invasive technique, usually under local anesthetic.
- Processing: The harvested material is processed to concentrate the stem cells or SVF. This step typically takes one to two hours at the clinic.
- Injection: The concentrated cells are injected directly into the affected joint, guided by ultrasound or fluoroscopy to ensure precise placement.
- Recovery: Most patients return to light activity within one to two days, though full effects may take four to eight weeks to become noticeable.
Pro Tip: Bone marrow cell procedures performed under ultrasound guidance tend to show more consistent outcomes in research studies. When evaluating providers, ask whether imaging guidance is a standard part of their protocol.
On the safety front, the evidence is reassuring. The Cochrane Systematic Review found that stem cell injections are generally safe for joints, with common mild adverse events including injection-site pain (relative risk 2.04 compared to placebo) and temporary swelling (relative risk 3.39). Importantly, serious adverse events were actually less frequent in the stem cell groups than in placebo groups (relative risk 0.72), and no tumor formation was reported across the reviewed studies.
| Treatment comparison | Pain improvement | Structural change | Serious adverse events |
|---|---|---|---|
| Stem cell therapy vs. placebo | Modest but significant | Not consistently observed | Lower than placebo |
| Stem cell therapy vs. PRP | Comparable | Similar (minimal) | Comparable |
| Stem cell therapy vs. corticosteroids | More sustained | Not observed | Generally lower long-term |
Effectiveness is best for people with moderate osteoarthritis who have not found adequate relief from physical therapy, oral medications, or corticosteroid injections. The evidence does not yet support stem cell therapy as a first-line treatment for early joint pain, nor as a disease-modifying intervention for severe bone-on-bone degeneration.
Nuances: Who benefits, edge cases, and what to ask your provider
With effectiveness and safety data covered, it’s important to dig into the subtleties and practical considerations before deciding.
Not everyone responds to stem cell therapy equally. Research published in Rheumatology International found that benefits are most consistent in older patients, when culture-expanded or bone-marrow-derived cells are used, and when the injection is guided by ultrasound. Repeated injections appear to enhance outcomes over time, though they also carry a higher rate of mild side effects like temporary swelling and soreness.
The patients most likely to benefit share several characteristics:
- Moderate osteoarthritis: Not early-stage (where lifestyle changes may suffice) and not end-stage (where total joint replacement may be unavoidable).
- Adults who have tried and not fully responded to conservative care: This includes physical therapy, NSAIDs (non-steroidal anti-inflammatory drugs), and corticosteroid or PRP or cortisone injections.
- People seeking to delay or avoid surgery: Stem cell therapy can be a meaningful bridge treatment, buying months or years of improved function before surgical intervention becomes necessary.
- Individuals without major systemic conditions: Active infections, blood disorders, or certain cancers may disqualify a patient from treatment.
Long-term data beyond two years is still limited. This is an important caveat. The current evidence supports improved symptoms over 6 to 12 months, with some patients maintaining benefits longer, but we cannot yet state with certainty how durable the effects are over five or ten years.
Before committing to treatment, here are the most important questions to ask your provider:
- What is the source of the stem cells, and are they autologous or allogeneic?
- Will imaging guidance (ultrasound or fluoroscopy) be used during injection?
- What is the cell dose, and is it within the range supported by clinical research?
- What outcomes are realistic for my specific degree of joint degeneration?
- What is the follow-up protocol if my symptoms do not improve within 8 to 12 weeks?
Pro Tip: A trustworthy provider will give you honest, research-based answers to these questions without overpromising dramatic results. Be cautious of anyone guaranteeing cartilage regrowth or cure-level outcomes.
The real story: What most people misunderstand about stem cell therapy for joints
We want to be candid with you, because we think you deserve a clear picture rather than a polished sales pitch.
The biggest misconception we see is that stem cell therapy will regrow cartilage. Patients often arrive hoping for a biological rebuild of their joint, imagining that damaged tissue will be replaced with fresh, healthy cartilage. The science tells a more nuanced story. As research confirms, stem cell therapy works primarily through anti-inflammatory and paracrine effects rather than robust cartilage regeneration. MRI scans after treatment rarely show meaningful structural changes, even when patients report significantly less pain and better function.
That distinction matters enormously. It means the therapy is a powerful symptom-modifying tool, not a cure. For many adults in North Texas managing daily joint pain, that distinction is actually very good news, because symptom relief and functional improvement are exactly what allow you to walk comfortably, keep up with your grandchildren, and stay active in a demanding Texas climate.
We also strongly believe that established therapies should come first. Physical therapy that strengthens the muscles surrounding a joint can reduce load on damaged cartilage significantly. PRP and cortisone injections have their own evidence base and are typically more accessible and less expensive. Stem cell therapy makes the most sense as a next step when those options haven’t delivered enough relief, not as a first-line treatment for someone who hasn’t yet tried a structured physical therapy program.
Our practical advice for North Texans choosing your therapy is this: approach stem cell therapy with clear, realistic goals. If your goal is to reduce daily pain by a meaningful margin, improve your walking tolerance, and buy time before considering surgery, stem cell therapy has a solid evidence base to support that goal. If your goal is complete pain elimination or a fully restored joint, the current evidence cannot promise that outcome.
Working with a provider who is transparent about what the research shows and what it doesn’t, is the single most important factor in having a positive experience with this treatment.
Explore your options for joint pain recovery
Armed with balanced information, you may be ready to take a meaningful step toward greater joint comfort and mobility. At Nortex Tissue Regeneration, we offer advanced stem cell therapy alongside PRP therapy for joints, giving you access to a full spectrum of regenerative options in one place. Our team of North Texas pain experts will review your imaging, discuss your history with conservative treatments, and help you build a personalized plan grounded in current clinical evidence. The conversation costs you nothing, and the clarity it brings could change how you approach your pain entirely. Reach out to us today and let’s talk through your options together.
Frequently asked questions
Is stem cell therapy for joints safe?
Clinical studies show stem cell joint injections are generally safe, with only mild side effects like temporary swelling and injection-site soreness, while serious adverse events are actually rarer than in placebo groups and no tumor formation has been reported.
Do stem cell injections rebuild cartilage?
While MSCs can theoretically differentiate into cartilage cells, most clinical benefits come from anti-inflammatory and paracrine effects rather than measurable cartilage regrowth, as MRI studies show no consistent structural changes after treatment.
How long do the effects of stem cell therapy last?
Most studies track outcomes over 6 to 12 months and show sustained symptom improvement within that window, though long-term data beyond two years is still limited and repeat injections may be needed to maintain results.
Who should consider stem cell therapy for joints?
Adults with moderate osteoarthritis who want a non-surgical option and have already tried physical therapy or injections are the best candidates for stem cell therapy, as it is a symptom-modifying treatment rather than a first-line or disease-curing intervention.
Recommended
- Stem Cell Therapy – Nortex Tissue Regeneration
- PRP vs Stem Cell Therapy: Which One Makes Sense for You? – Nortex Tissue Regeneration
- PRP vs Cortisone Shots: Which One Is Better for Joint Pain Relief? – Nortex Tissue Regeneration
- How Does PRP Compare to Prolotherapy for Joint Healing? – Nortex Tissue Regeneration
- Safe Non-Invasive Treatments: Holistic Options for Lasting Relief
- Prevenire la degenerazione articolare: strategie efficaci