Stem cells don’t work the way most marketing materials suggest. If you’ve been told that a single injection will regrow your worn cartilage within weeks, that claim doesn’t match what current research actually shows. The best-supported short-term healing mechanism for most stem cell therapies is paracrine signaling, meaning cells release bioactive molecules that reduce inflammation and support repair, rather than physically replacing damaged tissue. What that means for you as a patient dealing with knee pain, hip stiffness, or a stubborn sports injury is worth understanding clearly before you make any treatment decision.
Table of Contents
- How stem cells work inside your joints
- The real mechanism: Paracrine effects, not just ‘cell replacement’
- What real evidence shows: Outcomes, limitations, and safety
- Critical safety and regulation facts for North Texas patients
- Our view: The truth about stem cell healing most clinics won’t tell you
- Next steps: Explore safe, evidence-based tissue regeneration options
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Healing is gradual | Stem cell therapies mainly improve symptoms by supporting tissue repair, not instant tissue regeneration. |
| Mechanism is signaling | Most benefits come from paracrine effects—messenger molecules—not direct cell replacement. |
| Evidence is mixed | Clinical results show pain and functional improvements but only modest structural repair in most adults. |
| Safety requires scrutiny | Always verify FDA status and product quality to reduce risk from unapproved or poorly manufactured stem cell treatments. |
How stem cells work inside your joints
Your joints are not passive structures. They contain living, active populations of stem and progenitor cells that quietly maintain tissue health throughout your life. These cells sit in specific locations called niches, and stem/progenitor cells contribute to tissue repair by acting within synovial and cartilage-associated compartments in adult joints.
Understanding where these cells live helps explain why joint healing is gradual rather than sudden. The synovial lining, the thin membrane surrounding your joint, contains mesenchymal stem cells (MSCs) that respond to injury signals. The sublining layer holds a reserve population that can mobilize during inflammation. Cartilage itself has a limited supply of chondrocyte progenitors, which is one reason cartilage heals so poorly after injury compared to skin or muscle.

| Joint compartment | Resident repair cells | Primary repair role |
|---|---|---|
| Synovial lining | MSCs, fibroblast-like synoviocytes | Inflammation regulation, lubrication |
| Sublining layer | Perivascular MSCs | Mobilization during injury response |
| Cartilage surface zone | Chondrocyte progenitors | Limited local cartilage maintenance |
| Subchondral bone | Bone marrow MSCs | Structural support, signaling |
There is an important distinction between your body’s own (endogenous) stem cells and those introduced through injection (exogenous). Your endogenous cells already know the local environment and respond to it continuously. Injected cells, by contrast, face a foreign and often inflamed environment where long-term survival is uncertain. Most injected MSCs do not permanently engraft into joint tissue. Instead, they act temporarily, releasing signals that modify the local environment before they are cleared by the immune system.
This is not a flaw in the therapy. It is simply how the biology works. Understanding this reality helps you set realistic expectations when exploring stem cells and joint healing and when evaluating stem cell treatment options alongside alternatives like PRP.
“Repair in adult joints is a regulated, gradual process governed by cellular niches and signaling networks, not a switch that can be instantly flipped by any single therapy.”
The real mechanism: Paracrine effects, not just ‘cell replacement’
When stem cells help healing, they do it primarily through communication, not construction. Paracrine signaling means a cell releases molecules that act on neighboring cells, changing their behavior without the releasing cell physically becoming part of the tissue. This is the dominant mechanism behind most of the clinical benefits patients experience.
The best-supported healing mechanism for MSC-based therapies is paracrine signaling rather than direct tissue replacement. Here is what that looks like in practice inside a painful knee joint:
- Anti-inflammatory signals. MSCs release interleukin-10 and transforming growth factor-beta, which reduce the inflammatory cascade driving your pain and joint swelling.
- Growth factor release. Vascular endothelial growth factor (VEGF) and hepatocyte growth factor (HGF) support local tissue repair and blood vessel health.
- Immune modulation. MSCs can suppress overactive immune responses that accelerate cartilage breakdown in conditions like osteoarthritis.
- Extracellular vesicle secretion. Tiny packages called exosomes carry microRNA and proteins that reprogram local cells to behave in a more regenerative way.
- Chondroprotection. Signals from MSCs can slow the death of existing cartilage cells, preserving what you have rather than building new tissue.
This explains something that confuses many patients: why your pain and mobility can improve meaningfully even when a follow-up MRI shows no dramatic structural change. The tissue isn’t necessarily rebuilt, but the inflammatory environment that was driving your symptoms has been calmed. The stem cell secreted factors responsible for this effect are also relevant in PRP therapy, which works through a related but distinct set of growth factors.
Pro Tip: If a clinic promises that you will see new cartilage on your MRI after stem cell therapy, ask them to show you peer-reviewed trial data supporting that claim. Symptom relief is well-documented; structural regrowth on imaging is not.
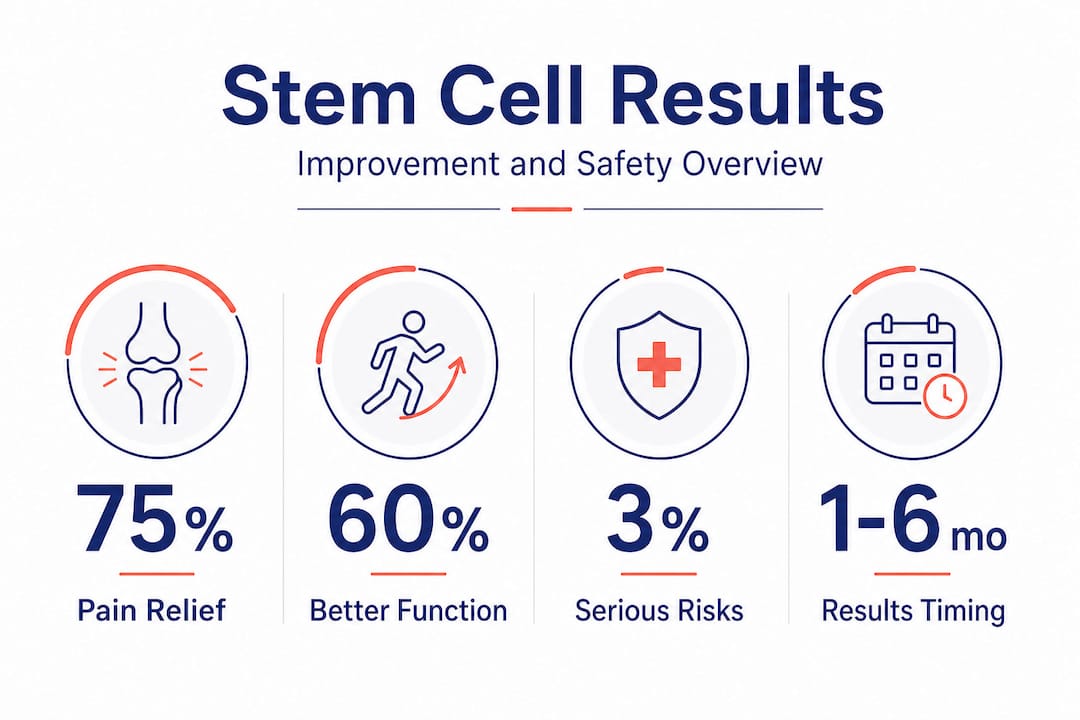
Clinical studies consistently show that patients report meaningful reductions in pain scores and improvements in function measures like walking distance and stair climbing. These are real, valuable outcomes. They just come from a different biological process than what the word “regeneration” implies in most advertising.
What real evidence shows: Outcomes, limitations, and safety
Understanding how stem cells help is one thing; seeing what the clinical evidence actually delivers for real patients is another. The research picture is genuinely promising in some areas and genuinely limited in others.
MSC therapy can improve pain and function in knee osteoarthritis, but evidence across trials is heterogeneous and effects are often symptomatic rather than consistently structure-modifying. In plain terms: many patients feel better, but their joints don’t necessarily look better on imaging.
Individual randomized trials show signals of benefit in selected osteoarthritis subgroups, particularly those with active synovitis, but also highlight the need for larger studies with longer follow-up periods.
| Outcome measure | What trials show | Confidence level |
|---|---|---|
| Pain reduction | Consistent improvement vs. baseline | Moderate to good |
| Functional improvement | Meaningful gains in most trials | Moderate |
| Cartilage regrowth on MRI | Generally non-significant changes | Low |
| Synovitis reduction | Improvement in subgroups at 6 months | Moderate |
| Long-term durability (2+ years) | Limited data available | Low |
The safety profile for properly prepared MSC injections is generally favorable. The most common side effects are local and temporary:
- Injection-site pain lasting 24 to 72 hours
- Temporary joint swelling or warmth
- Mild stiffness in the treated joint
- Occasional low-grade fever in the first 24 hours
Serious complications are rare but real. Infection, immune reactions, and in cases involving poorly regulated products, more severe systemic events have been reported. This is why product preparation quality and regulatory compliance matter so much.
Pro Tip: Ask your provider specifically whether the stem cell product being used is autologous (from your own body) or allogeneic (from a donor). Each carries different risk profiles and regulatory considerations that affect your safety.
For patients considering PRP and stem cell outcomes together, it is worth noting that combination protocols are being studied. Some early data suggests that PRP may enhance the environment for MSC activity, though this remains an active area of investigation. When comparing your options, reviewing the evidence for joint pain therapies side by side gives you a clearer picture of where each fits.
Critical safety and regulation facts for North Texas patients
Especially when exploring advanced options like stem cell therapy, understanding the regulatory landscape is not just wise, it is essential for your safety. The United States has a specific framework governing how cell-based therapies can be legally offered, and not every clinic operating in Texas follows it.
Many stem cell and cell-tissue products used outside FDA approval are regulated as drugs or biologics and may carry unverified quality, safety, purity, or potency risks. The FDA has issued formal warnings about clinics marketing unapproved products, including cases where patients experienced serious harm including blindness and tumor formation.
Safety and efficacy must be proven before marketing, and processing and manipulation quality is a critical determinant of risk, according to the International Society for Stem Cell Research. The way cells are harvested, processed, stored, and injected directly affects both their effectiveness and your safety.
Here is a practical checklist for evaluating any stem cell therapy provider in North Texas:
- Verify FDA regulatory status. Ask whether the product is FDA-cleared, approved, or falls under an Investigational New Drug (IND) application. Products that are “minimally manipulated” and used for “homologous use” may qualify under a different regulatory pathway, and your provider should be able to explain this clearly.
- Ask about cell source and processing. Autologous cells (your own bone marrow or fat tissue) carry lower immune risk. Allogeneic products from outside donors require more rigorous quality controls.
- Request documented clinical outcomes. A reputable provider should be able to share published or tracked outcome data, not just patient testimonials.
- Check for board-certified oversight. The physician directing your care should hold relevant credentials in orthopedics, sports medicine, or regenerative medicine.
- Avoid guarantees. No ethical provider can guarantee structural cartilage regrowth. If a clinic promises it, that is a warning sign.
“Patients deserve honest information about what stem cell therapies can and cannot do. Regulatory compliance and transparent outcome data are the foundation of trustworthy care.”
Pro Tip: You can search the FDA’s database of warning letters and enforcement actions online to check whether a specific clinic or product has received regulatory scrutiny. It takes five minutes and could protect your health.
Reviewing what stem cell product safety standards look like in a compliant clinical setting gives you a useful benchmark. Understanding FDA and patient safety considerations in the context of biologics helps you ask better questions before committing to any treatment plan.
Our view: The truth about stem cell healing most clinics won’t tell you
We want to be direct with you about something most promotional materials in regenerative medicine avoid saying out loud: the gap between marketing language and clinical reality is wide, and it does not serve patients.
The word “regeneration” implies rebuilding. It suggests that damaged tissue is restored to its original state, the way a broken bone heals back to full strength. For most adult joints treated with current stem cell therapies, that is not what happens. What does happen, often, is meaningful symptom relief driven by paracrine signaling, inflammation reduction, and immune modulation. That is genuinely valuable. But it is a different thing.
We see patients who have been told by other providers that they will grow new cartilage, that their joint will be “like new,” or that they can avoid surgery indefinitely with a single injection. When those promises don’t materialize, patients lose trust in regenerative medicine as a whole, and that is a real loss because the honest version of what these therapies offer is still worth considering for many people.
Because regulation and product quality vary widely across clinics, US patients should be cautious with marketing claims and ensure any stem cell therapy aligns with lawful FDA-regulated status and evidence. This isn’t bureaucratic caution. It’s the difference between a therapy that helps you and one that harms you.
The questions we think every patient should ask before starting stem cell therapy are simple: What is the source of the cells? What does your outcome data actually show? What happens if I don’t improve? A clinic that answers these questions clearly and honestly is one worth trusting. One that deflects or offers guarantees is one worth leaving.
Our approach at Nortex is built on science-based stem cell care that prioritizes your informed consent and realistic expectations. We believe that a patient who understands the real biology makes better decisions and gets better outcomes.
Next steps: Explore safe, evidence-based tissue regeneration options
If this article has clarified what stem cell therapy can realistically offer, and raised questions about whether it’s the right fit for your specific condition, that is exactly where we want you to be. Informed patients are better patients.
At Nortex Tissue Regeneration, we build every treatment plan around your individual anatomy, diagnosis, and goals. Our process for Nortex stem cell therapy includes a thorough evaluation of your candidacy based on current clinical evidence, transparent discussion of cell sourcing and FDA alignment, and honest outcome tracking so you know what is actually working. We serve patients across North Texas who are ready to move beyond chronic pain without rushing into surgery.
Visit Texas Tissue Regeneration to learn more about our clinic and care philosophy, or explore our full range of regenerative medicine options to compare therapies and find the right starting point for your recovery.
Frequently asked questions
Do stem cell treatments regrow cartilage in damaged joints?
Most stem cell treatments do not regrow cartilage visibly on MRI, but they can relieve pain and improve function for many patients. MRI structural outcomes were non-significant in key trials, even when patients reported meaningful pain and functional improvements.
Are stem cell therapies for joint pain FDA approved?
Few stem cell therapies for joints are FDA approved, and unapproved products may pose serious risks to your health. FDA-regulated unapproved products from human cells or tissues may carry unverified safety, purity, and potency risks that patients are rarely warned about.
How long does it take to see results from stem cell therapy?
Most patients notice changes in pain or function within one to six months, but full structural healing is slow and not guaranteed for every patient. Significant synovitis improvement at 6 months has been documented in trials, while broader structural MRI changes were not consistently demonstrated.
What are the possible side effects of stem cell joint treatments?
The most common side effects are temporary injection-site pain and joint swelling that resolve within a few days. Higher local reaction rates and uncommon serious complications have been documented in clinical trials, with serious events being rare in properly regulated settings.
How do I know a clinic’s stem cell therapy is trustworthy?
Ask directly for FDA approval or regulatory status, documented cell source and handling processes, and published or tracked clinical results. Regulators and expert bodies emphasize that safety and efficacy must be proven before marketing, and that contamination and process quality are critical risk factors patients should understand before consenting to treatment.